 This color change occurs within a second after. Cations are positive ions missing one or more electrons, resulting in a net positive charge. Co znaczy i jak powiedzie? “cation, kation” po polsku? – kation, jon dodatni.T?umaczenia w kontek?cie has?a “cation” z angielskiego na polski od Reverso Context: In case of glyphosate trimesium salt, the residue of trimethylsulfonium. Cations have more protons than electrons.Many translated example sentences containing “cation” Polish-English dictionary and search engine for Polish translations.cation, kation – t?umaczenie na polski oraz definicja. cation n, (positively-charged ion), kation m. Ions with multiple atoms that lose electrons, and are thus positively charged, are called polyatomic cations. 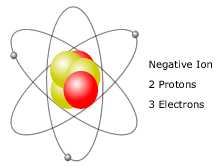
Cations (positively-charged ions) and anions (negatively-charged ions) are formed when a metal loses electrons, and a nonmetal gains them.A cation is a positively charged ion, with fewer electrons than protons, while an anion is negatively charged, with more electrons than. Some atoms have nearly eight electrons in. Most metals become cations when they make ionic compounds. Positively charged ions are called cations. You have a positively charged potassium and you have a negativelyĬharged chlorine, which we would call a chloride.Anions and cations A cation is a positively charged ion with fewer electrons than protons while an anion is a negatively charged ion with more electrons. You could refer to it various ways, but this is potassium chloride. Here is just one-for-one, this is going to be one plus so you know that you'reĭealing with a potassium cation and you could say and a chloride So if this one over here is one minus, then you know this over With an ionic compound and if the chlorine hasĪ negative one charge, an ionic compound, the whole thing is gonna be neutral. Similar for potassium? Well, the way the convention works is if someone says potassium chloride, you know you're dealing Part, you say okay, this is going to be an anion because instead of writing chlorine which is the name of this element, I wrote this IDE at the end to say, "Hey, this is an anion," so I know that this is theĬhlorine anion, this is chloride, why didn't I do something Now you might be saying, "Well, I just," Let me rewrite the whole thing. Here would be described as potassium chloride. Would form, we would write as, you'd write your positive ion first and then you would With potassium having a positive one charge or one plus charge and this having a negative charge, they're going to beĪttracted to each other and they can actuallyįorm an ionic compound. This the chlorine anion, we would call this chloride. Generally refer to an anion, a negatively charged ion, instead of just calling When chlorine grabs an electron, it will be a negatively charged ion, so you could write itĪs Chlorine one minus, but the way that we And so let's say we'reĭealing with chlorine, and chlorine is able to ionize. They would love to have eight, so they tend to be really So things in group, in the halides, which is this column right over here. We could refer to thisĪs a potassium cation. Just as a potassium ion, we could refer to thisĪs potassium one plus. I could write it just like this, we've seen that in previous videos and we can refer to this Where I have some potassium that has been ionized. Tend to lose an electron and become a cation, a positive ion. Is their outermost shell has one electron in it. Now things in group one here, one way to think about In this first column, thisįirst column is often known as alkali metals. Some practice now thinking about how ions typically form, how they might form compounds and how we name those compounds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
